AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Neo melubrina8/22/2023 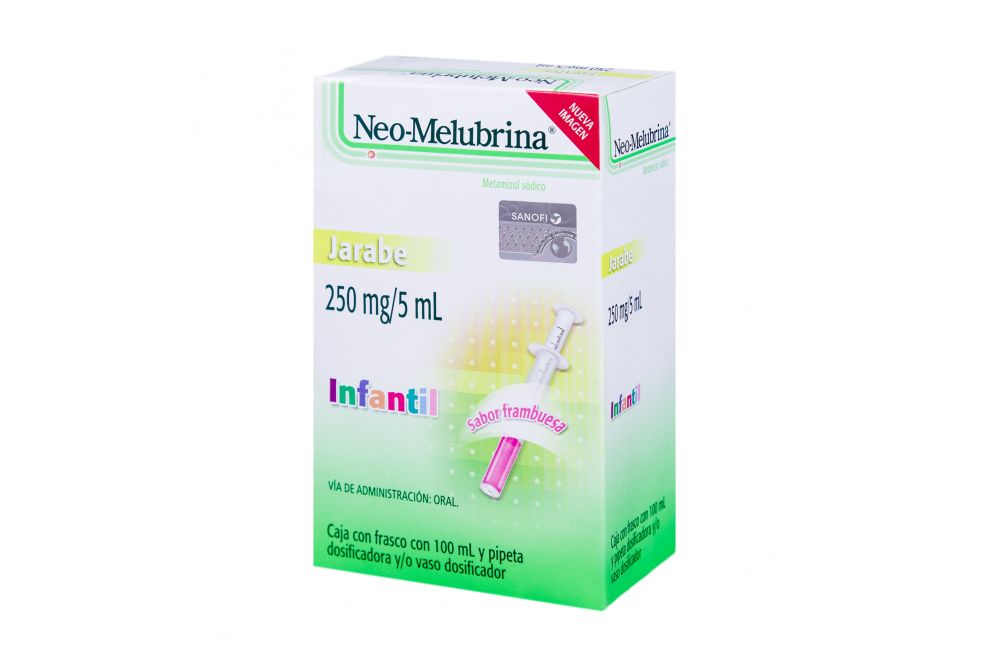
As part of the FDA's high standards, drugs can be manufactured only at plants registered with the agency, whether those facilities are domestic or foreign. Interstate shipment includes importation-bringing drugs from a foreign country into the United States.ĭrugs sold in the United States also must have proper labeling that conforms with the FDA's requirements, and must be made in accordance with good manufacturing practices. Under the FD&C Act, the interstate shipment of any prescription drug that lacks required FDA approval is illegal. 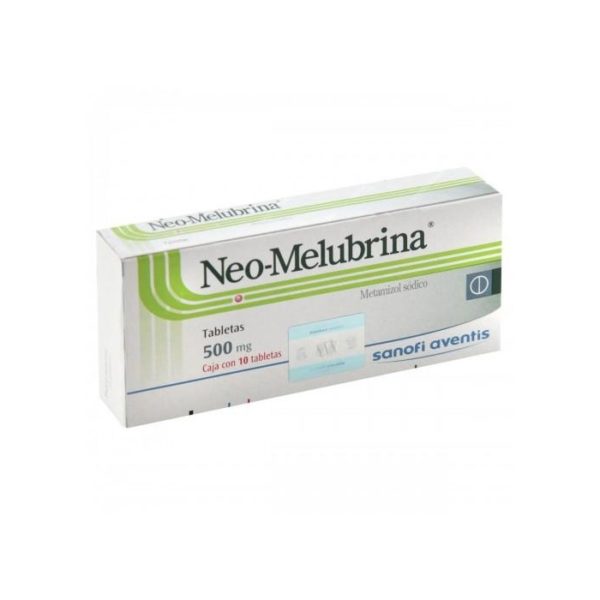
Joe McCallion, a consumer safety officer in the FDA's Office of Regulatory Affairs, sums it up this way: "If you buy drugs that come from outside the U.S., the FDA doesn't know what you're getting, which means safety can't be assured." Benefits of a Closed System Drug standards and regulations vary from country to country, and the FDA is responsible only for those marketed and sold inside the United States. And just because a drug is approved in a foreign country, that doesn't mean it's approved in the United States. Whether you're searching for a cheaper price or dodging the doctor's office, the FDA warns against using unapproved drugs. And if you experience some kind of allergic reaction or other side effect, it's hard to trace the problem and treat it." "The drugs could be old, contaminated, or counterfeit. "I've seen eye medications that look like they're 20 years old," Hancz says. The list of safety risks is long, but the principal problems involve the use of prescription drugs without a physician's supervision and the danger of buying drugs of unknown origin and quality. Attorney's Office for the Central District of California has indicated that legitimate or state-licensed clinics exist where immigrants can be treated safely regardless of immigration status. Hancz says that HALT has seized prescription drugs found mostly in Latino, Asian, and Russian immigrant communities, where some undocumented immigrants, fearing that their immigration status may be discovered, have sought health care in back rooms. Other drug sellers have taken their businesses underground, moving from storefronts to private homes in an attempt to hide.Īs in Selene's case, some criminals have falsely claimed to have a medical background and not only illegally sold drugs, but administered injections. 
Local lawmakers have stiffened penalties, and many illegal pharmacies have been shut down. Ring members have been arrested and accused of violating the Federal Food, Drug, and Cosmetic Act (FD&C Act). The FDA's Office of Criminal Investigations in Los Angeles has teamed with HALT to uncover major black market pharmacy rings selling Spanish-labeled pharmaceuticals. "Then there are controlled substances like Valium, which you do need a prescription for in Mexico." "Plenty of drugs that require a prescription in the United States-like antibiotics, cardiac drugs, and birth control pills-are available over the counter in Mexico," he says. Marv Shepherd, Ph.D., director of the Pharmacoeconomic Center at the University of Texas at Austin, places drugs available in Mexico into two categories. Experts say the problem mirrors what goes on in nearby Mexico, where easy access to prescription drugs is common. HALT was launched as part of the crackdown, and task force members have confiscated a variety of prescription drugs being sold illegally. "We've found drugs that were stored in tin containers and car trunks," says Daniel Hancz, Pharm.D., a pharmacist with the Health Authority Law Enforcement Task Force (HALT) in Los Angeles, an organization of police officers and other law enforcement personnel with special training in pharmaceuticals. Her death set off a crackdown in December 2000 on smuggling drugs from Mexico and selling them at swap meets, gift stores, clothing stores, meat markets, and other retail establishments in Southern California. That was 20 years after the Food and Drug Administration had banned the drug in the United States because of potentially fatal side effects, including a drop in white blood cells that hampers the body's ability to fight off infections. Selene Seguros Rios was 18 months old in 1999 when she received two injections of a pain and fever drug called Neo-Melubrina (dipyrone) in an illegal backroom clinic in Tustin, Calif.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
